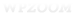New Developments in Magnetic Bead Immunoassays
Functionalized magnetic beads are widely used for diagnostic assays and cell isolation. These micro- or nano-beads are made from a paramagnetic material, commonly magnetite (Fe3O4) that is not innately magnetic, but becomes magnetized when placed into a magnetic field. When purchasing these beads one can select from a variety of surface coating platforms that provide chemical reaction nodes for covalent or non-covalent bonding of desired antibodies or ligands. The governable specificity of antibody binding transforms this technology into a powerful immunoassay. Of course, the magnetic beads are not the only part of the story. One cannot underestimate the importance of a properly scaled magnetic separation system. Coupling magnetic beads with a target cells is useless without a way to isolate the conjugates for further study.
The basic protocol of magnetic separation is deceptively simple. First, determine the target molecule or cell. Second, purchase a magnetic bead that can be functionalized with an antibody or ligand specific to your target. Third, purchase a magnetic separation system to isolate the target from solution. Fourth, recognize the potential for fine-tuning each of those steps. Some areas of advancement to consider include:
- Control over bead coatings and surface functionalization
- Tailoring bead size to the experimental goal
- Scalability of the magnetic separation system
Bead Coatings and Surface Functionalization
In the past few years the magnetic separation community has seen a rapid improvement in control of functionalization chemistry. Originally, ligands were attached to bare magnetic beads by simple passive adsorption of molecules to the bead surface, which relies on weak hydrophobic interactions to associate ligands to a bare bead. It works, but it often results in unreliable and non-specific binding affording little control over ligand orientation or stability.
New developments have introduced modified bead surfaces (carboxyl, amino, hydroxyl, and sulfates), pre-activated surfaces (tosyl, epoxy, and chloromethyl groups), and bio-activated surfaces (protein A, protein G, streptavadin-biotin). Firstly, modified bead surfaces provide a way to covalently attach surface antibodies, and can be quite useful. However, functionalization of these surfaces must be done in a non-polar solvent and aggregation and non-specific binding can be an issue. Many of the sites are common to multiple proteins and locations within a single protein.
Pre-activated bead surfaces such as tosyl, epoxy, and chloromethyl groups add a level of control through manipulation of solution pH. Tosyl actively binds to sulfhydryl groups at a neutral pH, but switches to amino groups as the solution becomes more basic.
The most expensive, but most specific option is to use bio-activated surfaces of protein A/G and streptavadin-biotin affinity ligands. Streptavadin has an incredible specificity for biotin, and the affinity between the two is unaffected by changes in pH, salt concentration, or the presence of detergents. This specificity can be very useful. A notable feature of this system is that the link between streptavidin and biotin is reversible when exposed to a short 70°C incubation. This provides an easy way to separate and remove magnetic beads after target isolation.
All of these surface functionalization methods provide control over specificity and stability of the bead-target coupling, but they don’t address functional group orientation or density. These strategies all require incubation of the functional moieties in molar excess with the magnetic beads. This can result in the attachment of functional groups upside-down or at angles that decrease target-binding efficacy. Similarly, such excess creates a high density of functional groups in multiple layers on one bead, and steric effects can block target-binding access. Current and future solutions to this problem have arrived in the form of improved protocols for magnetic bead functionalization, a deeper understanding of the kinetics of antigen capture, and development of new conjugation chemistry. This new conjugation chemistry consists of new polymeric metals ions and coordination complexes to improve control over ligand density and orientation on a variety of surfaces. More detailed information and references to this research can be found within the SEPMAG ebook entitled Magnetic Bead Coatings Today and Tomorrow.
Tailoring bead size to the experimental goal
The bead size affects the physical and chemical aspects of the magnetic separation process. Specific area is defined as the amount of binding surface divided by bead mass. This can also be thought of as binding area per gram of material. Smaller beads have a greater specific area, which is beneficial for reducing the cost of support material for the protein. However, smaller beads take more time to move along a magnetic field gradient. Thus, a balance between binding capacity and separation time must be optimized for each experiment.
It is possible to increase the separation speed by increasing the magnetic content of the beads, but attention must be given to the density. Increasing the density too much would cause the beads to precipitate out of solution before they are collected by magnetic separation. Magnetic separation is conducted horizontally so the beads travel to the sides of the container, but if the beads are too dense they will succumb to gravity and form a sediment at the bottom. Another potential problem with increasing the magnetic content too much is that it can increase the magnetic force on the beads, which could increase inter-bead interactions and cause irreversible aggregation.
Regarding bead size, another important factor to consider is the size distribution of the batch. Manufacturing methods vary in the amount of control afforded over the uniformity of bead size in the batch. A large size distribution, meaning the beads are not uniform in size, could result in a high degree of variability in the separation process. This could be problematic for highly sensitive assays that demand consistency in the final product. When purchasing beads it is worth taking the time to consider and compare size distribution data in order to ensure that the bead size is uniform enough to achieve a consistent magnetic separation and a good result.
Scalability of the magnetic separation system
All of the surface coating technology and bead manufacturing techniques will mean nothing without a proper magnetic system to recover them from solution. Proper means that the separation rack is not just tailored to the sample size, but has well defined working conditions to ensure that all bead-target conjugates are recovered. Classical separation racks generate a magnetic force that changes with distance from the retention area. The problem with this design is that for having enough magnetic force to collect the beads farthest from the retention area, the force over the beads closest to retention would be extremely high. Excessive force generates irreversible aggregates closest to the retention area, especially if the separation time is large due the slow separation speed of the beads farthest away. This lack of consistency may be confusing, and troubleshooters may erroneously look for problems with the beads or coating process. Much time and money can be wasted this way when the problem is really a result of the inhomogeneous force from a poorly designed separation device.The retention force must be uniform at any distance away from the retention area so that it can affect the farthest beads and avoid losses or very large separation times. Advanced biomagnetic separation systems generate a magnetic force that is homogenous over the working volume (i.e. the force is constant regardless the distance to the retention area). That provides a larger force over the farthest beads, avoiding losses and speeding up the separation process as well as avoiding irreversible aggregation. These features open the door to multiple washings without losing material. Moreover, as all the beads experience the same force during the same time, the consistency is extremely high.
New products such as those sold by SEPMAG ensure a gentle homogeneous magnetic field is applied during the separation process, but also incorporates real-time monitoring of the process. Thus, different process can be compared, not just by separation time, but also by dynamic feedback. Standard separation curves are used to calibrate the separation process depending on changes in concentration, bead size, and bead size distribution. These standard curves can be used to guarantee the quality of the process (key for batch-to-batch consistency), and also as powerful R&D tools to determine how protocol changes affect the separation process.
Conclusion
When designing a magnetic separation strategy for an experiment it is beneficial to optimize each of the major three steps in the process. This planning time will result in a rapid, efficient, and reliable separation process that is finely-tuned for the experiment at hand. The future is bright with developments making it easier than ever to transition to a magnetic-bead-based immunoassay.
Reference: SEPMAG ebook ‘Magnetic Bead Coatings Today and Tomorrow‘